ALBA Synchrotron
XAIRA beamline reached another milestone, with the first deposition of a new macromolecular structure in the Protein Data Bank determined from diffraction data obtained at XAIRA. The deposition of the atomic structure of this protein complex represents an early result of the beamline’s recently launched user program, with many more structures expected to follow.
The microfocus macromolecular crystallography beamline XAIRA achieved an important milestone this year by entering regular user operation in June. Since opening its doors to the structural biology community, XAIRA has rapidly demonstrated its value as a high-performance instrument for challenging crystallographic projects. In just a few months of activity, in parallel with the last phases of beamline commissioning, nearly 80 experimental shifts have been delivered to users, who have collected diffraction data from a wide range of crystal samples. This successful start reflects the confidence of users in XAIRA as a reliable beamline that enables their cutting-edge structural research.
A particularly significant achievement has taken place last week, when a new protein structure determined from data collected at XAIRA was deposited in the Protein Data Bank (PDB). The structure, deposited under the PDB identifier 9TJX, represents the first PDB entry derived from user data collected at the XAIRA beamline. Although the structure is on hold and thus not yet publicly released, the scientific article describing it is expected to be published in the near future, marking a key scientific outcome of XAIRA’s early operational phase.
The main author of this deposition is Jacinto López Sagaseta, Director of the Unidad de Cristalografía de Proteínas e Inmunología Estructural at Navarrabiomed (Pamplona). His group has long-standing expertise in structural studies of proteins involved in haemostasis and immune regulation, and this new structure addresses a long-standing question in the field of coagulation biology.
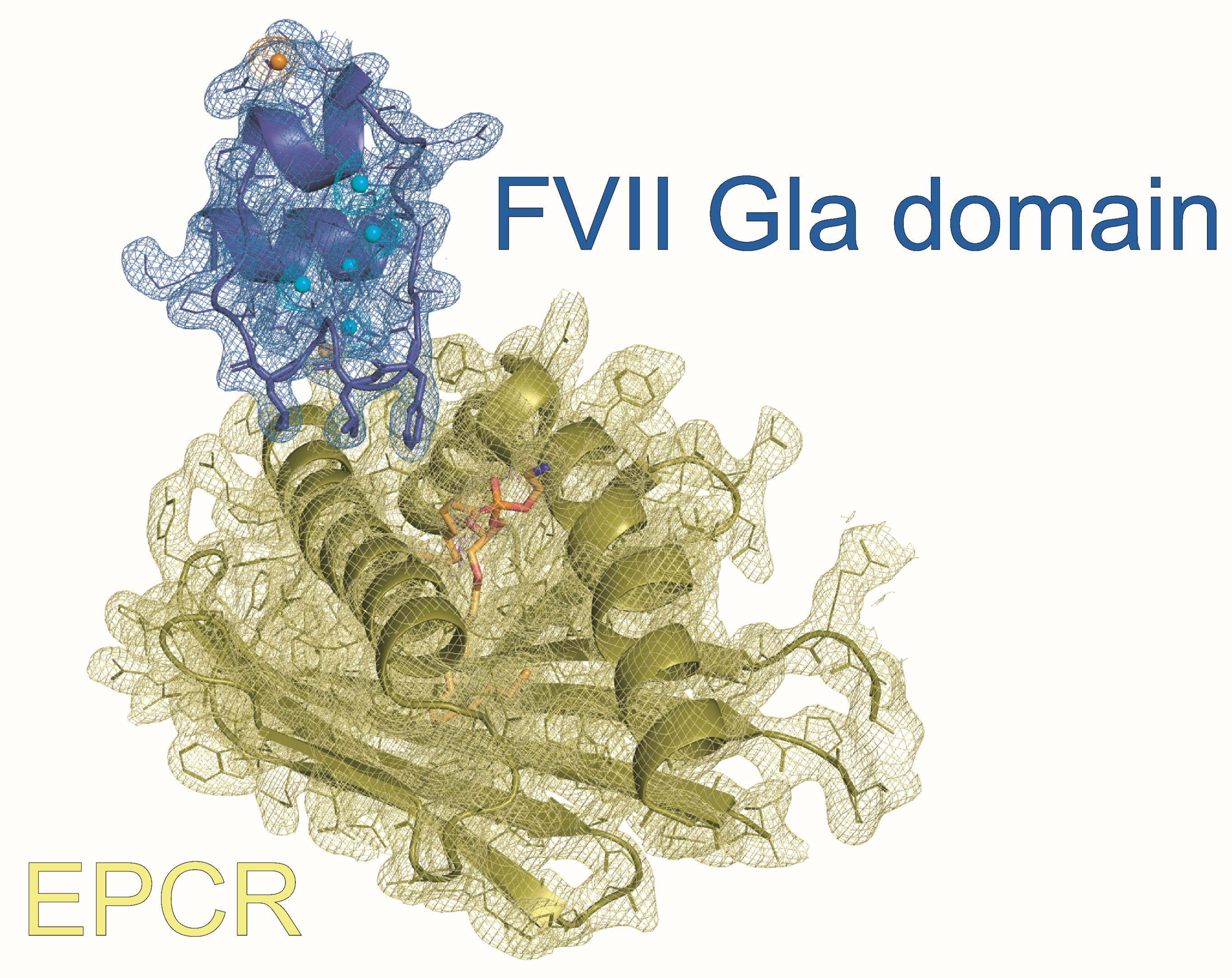
The study addresses the structural basis of the interaction between recombinant activated factor VII (rFVIIa), a key therapeutic agent for controlling severe bleeding in haemophilic patients, and the endothelial protein C receptor (EPCR). Although EPCR was known to bind FVIIa, the lack of structural information had limited understanding of the molecular mechanism underlying this interaction and its physiological and pharmacological relevance during rFVIIa treatment.
Using X-ray crystallography, the authors determined the structure of the FVII Gla domain bound to EPCR, revealing that FVIIa and protein C compete for the same binding site on EPCR through a conserved, Gla domain–dependent docking mechanism. This finding establishes EPCR as a genuine receptor for FVIIa, overturns the view that EPCR is restricted to protein C/APC binding, and provides a structural explanation for how rFVIIa may influence protein C activation during haemophilia therapy.
XAIRA beamline, with its highly focused X-ray beam of 4 × 6 µm² (expected to reach its nominal size of 3 × 1 µm² after further commissioning) and its low-noise high-frame rate Eiger2 XE 9M detector, is specially designed to enable efficient data collection from very small or inhomogeneous crystals. Remarkably, XAIRA is planned to move into operation in a helium atmosphere early next year, which will further reduce background scattering and endowing cleaner diffraction images from microcrystals and weakly diffracting samples, and will implement the fixed-target serial crystallography setup for room temperature experiments by mid next-year.
Beyond its biological significance, this first PDB deposition based on XAIRA user data stands as clear evidence to the beamline’s capabilities and scientific impact. As XAIRA continues to support an expanding user community, further high-quality structures and important biological insights are expected to follow, reinforcing its role as a key instrument for the structural biology community in Spain and abroad.