ALBA Synchrotron
Researchers have unveiled a new type of ultrathin cobalt–selenium nanosheet capable of driving the oxygen evolution reaction with unprecedented efficiency. This reaction is a key step in clean energy technologies. Now, the study uncovers how tuning the atomic structure of these materials can switch the fundamental reaction pathway by which oxygen is formed, offering a promising route to designing the next generation of water-splitting catalysts and high-performance zinc–air batteries. Experiments at CLAESS and MIRAS beamlines of ALBA were key to analyse how the catalyst operates under working conditions.
Oxygen evolution is essential for green hydrogen production and crucial in devices such as water electrolysers and rechargeable metal–air batteries. However, the oxygen evolution reaction (OER) is slow and energy-intensive. While cobalt hydroxide has long been studied as a low-cost alternative to noble-metal catalysts, it still struggles with limited efficiency and long-term durability.
Now, a collaborative team, which involves scientist from the Catalonia Institute for Energy Research (IREC), the ALBA Synchrotron, the University of Barcelona, and the Universities of Jinan and Xiamen in China, has developed an ultrathin cobalt–selenium (CoSe) catalyst that significantly accelerates the OER, a key bottleneck in renewable energy technologies.
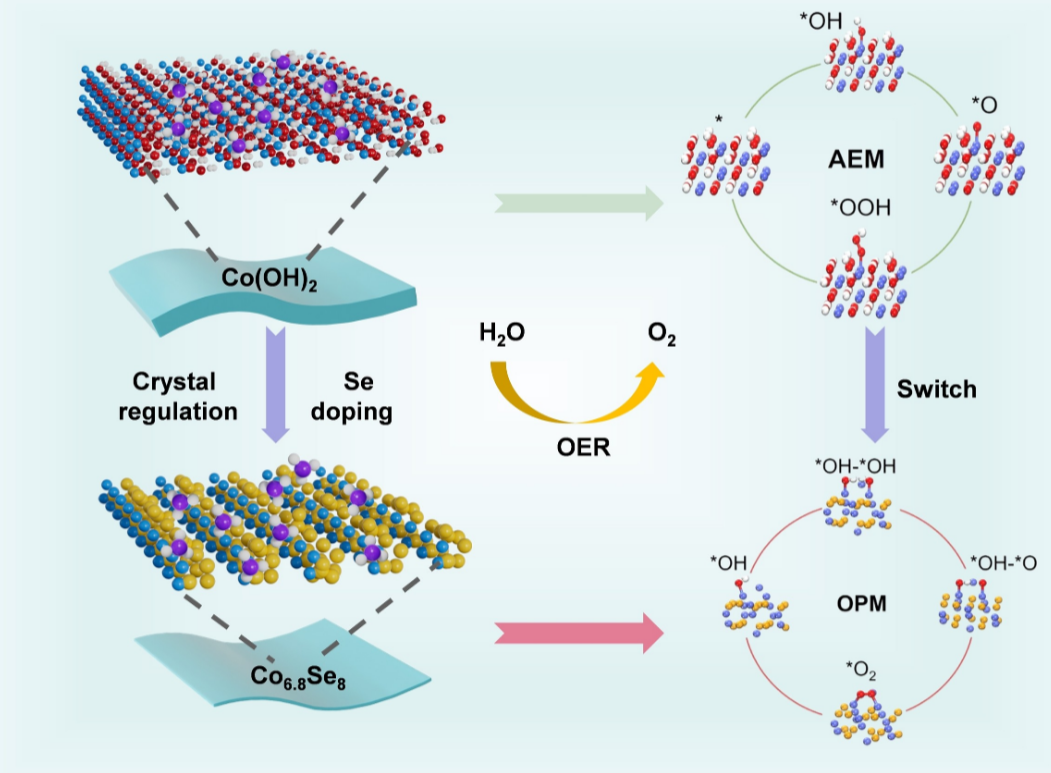
The research team approached the challenge by engineering a new family of ultrathin nanosheets — just one to two nanometres thick — made by inserting selenium into cobalt hydroxide. As the selenium content increased, the nanosheets changed composition, thickness, and atomic arrangement, forming the Co₆₈Se₈ phase. Among all the synthesized materials, this ultrathin phase proved to be the most active.
Though selenium itself does not catalyse OER, its incorporation dramatically reshaped the surface environment, and the cobalt local environment, altering how the metal interacts with oxygen-containing species. This subtle atomic engineering turned out to be essential for boosting the catalyst’s overall reactivity. Indeed, the results revealed that Co6.8Se8 employs a different reaction mechanism respect Co(OH)2 with a reduced cobalt species as the active sites.
A new mechanism enabled by surface reconstruction
The oxygen evolution reaction can follow different reaction pathways. The most common, the adsorbate evolution mechanism (AEM), involves the formation of an *OOH intermediate that requires substantial energy. This is one of the factors that slows down the reaction.
Thanks to both ex-situ and operando synchrotron techniques carried out at the ALBA Synchrotron, the researchers found that Co₆.₈Se₈ does not follow this conventional route. In particular, while Co(OH)2 follows the adsorbate evolution mechanism (AEM) pathway for OER with Co3+, Co6.8Se8 employs a different mechanism with Co2+ as the active sites.
Under working conditions, the catalyst’s surface partially reconstructs into a selenium-doped cobalt hydroxide layer that instead favours the oxide path mechanism (OPM). In this mechanism, oxygen radicals on the surface couple directly to form O₂, bypassing the energy-demanding OOH step.
On one hand, using the synchrotron-based FTIR at MIRAS beamline, the team monitored the infrared signatures of reaction intermediates in real time. They observed that oxygen species begin to form at lower voltages on the Co₆.₈Se₈ nanosheets than on pure cobalt hydroxide, and they detected signals characteristic of direct O–O radical coupling. That is clear evidence of the OPM pathway. The *OOH related to the conventional AEM pathways were not detected on the Co₆.₈Se₈ nanosheets. Unwanted side intermediates, such as those associated with hydrogen peroxide, were less prominent, indicating a more selective and efficient reaction.
On the other hand, X-ray absorption spectroscopy at the CLÆSS beamline provided atom-level structural information. These experiments revealed cobalt reduction, shorter cobalt–cobalt distances, and robust cobalt–selenium bonds in the ultrathin phase. Suitable Co-Co bond length facilitates the radical *O-*O coupling step of the OPM mechanism. Even after extended operation, selenium remained at the surface as protective SeOx species, preventing the cobalt from over-oxidizing and helping preserve activity.
Together, the experiments performed at ALBA linked the nanoscale structure of the material with its enhanced macroscopic performance, offering a clear explanation for its superior catalytic behaviour. By integrating synchrotron results with theoretical modelling, the researchers built a detailed mechanistic picture that guides how future catalysts might be engineered.
Strong performance and real technological potential
Electrochemical testing confirmed that Co₆.₈Se₈ is a high-performing catalyst. It requires only 253 mV to drive OER at 10 mA/cm² — one of the lowest overpotentials reported for cobalt-based systems. The nanosheets also showed rapid reaction kinetics and remarkable stability during operation.
To demonstrate the material’s practical relevance, the team incorporated the catalyst into rechargeable zinc–air batteries (ZABs), a promising technology for large-scale energy storage and portable electronics. In these devices, OER governs the charging process, and improving its efficiency can significantly reduce energy losses.
ZABs equipped with Co₆.₈Se₈ nanosheets achieved a higher open-circuit voltage than batteries made with pristine cobalt hydroxide, a power density near 80 mW/cm², a high specific discharge capacity, and outstanding durability over more than 500 hours and 1,500 charge–discharge cycles.
When combined with a commercial Pt/C catalyst, performance improved even further, showing that the new material can complement existing state-of-the-art electrodes.
Next-generation energy materials
Beyond cobalt selenide, the findings point to a general strategy: by precisely controlling growth and adjusting the distance at the atomic scale, and stabilizing specific surface configurations, it may be possible to trigger efficient O–O coupling mechanisms in other transition-metal materials. This approach could accelerate the creation of affordable, efficient catalysts for hydrogen production and advanced battery systems.

(a) ZAB schematic diagram. (b) The OCP value curves over time (inset: optical image of the ZAB), (c) power density, (d) specific capacity comparisons, and (e) galvanostatic discharge-charge curves with 10 min discharge and 10 min charge cycles at a current density of 4 mA/cm2 for a Co6.8Se8-based ZAB and a Co(OH)2-based ZAB. (f) Normalized XANES spectra at the Co K-edge of Co(OH)2, Co6.8Se8, and reference samples. (g) Relationships between valence states and positions of the first derivative maxim of XANES spectra at the Co K-edge. (h) FITR spectra.